This page includes details of three case studies where the influence of metal bioavailability on EQS compliance (calculated using the bio-met bioavailability tool v3) has using has been investigated. Each of these case studies was undertaken using readily available data and should not be interpreted as official examples of regulatory compliance in these Member States.
Introduction
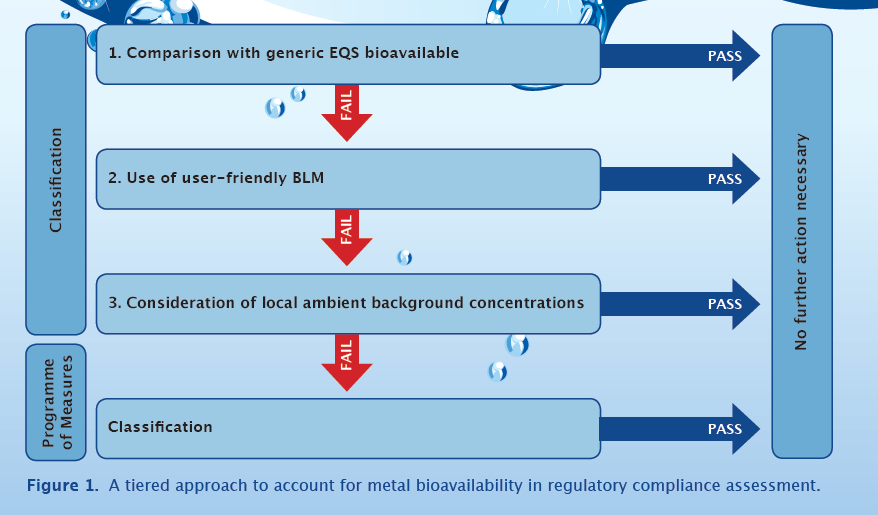
Accounting for the bioavailability of metals in routine water monitoring represents a step-change in the way regulators assess the potential risks of metals in the aquatic environment. Guidance on how bioavailability may be incorporated into compliance assessments, classification and local risk assessment are included in the recent EU Technical Guidance for deriving Environmental Quality Standards (EQS) under the European Water Framework Directive (WFD). The guidance supports the use of a tiered compliance assessment regime for metals (Figure 1) that incorporates bioavailability and recommends using Biotic Ligand Models (BLMs). However, a reasonable question remains before widespread adoption of this new approach: “How will the pattern of EQS differ from conventional assessments when bioavailability is taken into account?”
Tiered approach for compliance assessment
To determine compliance with an EQS regulators will usually monitor the metal concentration at a site over one a period of at least a year. The mean dissolved metal concentration is compared to the generic EQSbioavailable. Some European Member States will also assess the “confidence of failure” as this stage using guidance given in ISO/WD 5667-20. If the EQS is exceeded the assessment passes to tier 2 where site-specific pH, dissolved calcium and dissolved organic carbon (DOC) data are used in the bio-met bioavailability tool to account for site-specific bioavailability.
The outputs of these calculations include a local bioavailable metal concentration (which can be compared to the EQSbioavailable), a local EQSdissolved (which can be compared to local dissolved concentrations of metals) and also a local risk characterisation ratio (values > 1 indicate a potential risk at a site). As per stage 1, some Member States in Europe will assess the “confidence in failure” at tier 2. If the EQS is exceeded the assessment passes to tier 3.Tier 3 allows for local issues, including ambient background concentrations, to be taken into account.
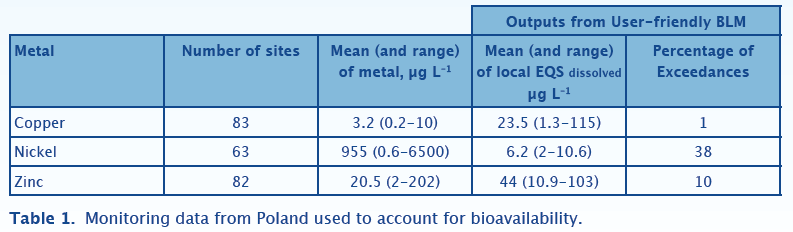
Polish Case Study
Table 1 shows the compliance assessment results for Polish monitoring data for the years 2008 and 2009 obtained from European Environment Agency’s website. There are relatively few sites, but each site has been sampled between 8 and 12 times over the period. The number of metals monitored at each site varies with fewer sites recording dissolved nickel. The sites in this dataset have relatively high dissolved organic carbon (median = 7.6 mg/l) and pH (mean = 7.9). However, the metal exposures, especially nickel, are very high. The outputs from the bio-met bioavailability tool show relatively few EQS failures for copper and zinc despite the high exposures. These data show the importance of dissolved organic carbon in mitigating the effects copper and zinc toxicity. For nickel the relatively high metal exposures result in EQSbioavailable failures of 38%, despite accounting for bioavailability. It is important to stress that it is not clear, from the website, whether the monitoring data are for dissolved or total metals.
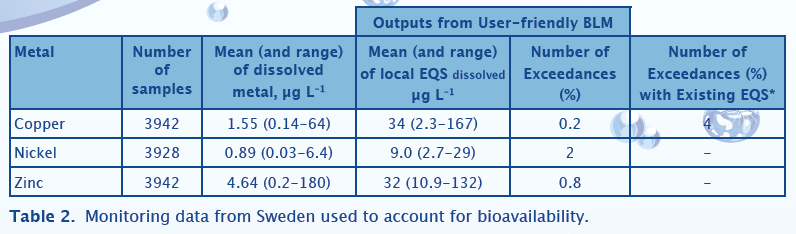
Swedish Case Study
Table 2 shows the compliance assessment results for a series of Swedish rivers and lakes. These data were made directly available from KEMI and cover the period 2000-2008. These data are not annual averages at sites, but represent a ‘face value’ assessment with individual samples, which is more precautionary than a conventional compliance assessment against annual average concentrations. For copper an additional comparison is made between failure rates observed using the existing Swedish EQS with the bio-met bioavailability tool. These data show that accounting for bioavailability results in fewer EQS failures. The waters in this dataset have a mean pH of 7.1, but a median dissolved organic carbon of 8.4 mg/l. This means these waters are relatively insensitive to exposures of copper, nickel and zinc which is supported by the low levels of EQS failure observed after bioavailability is accounted for.
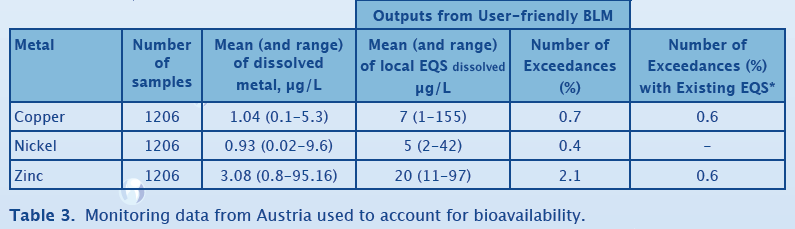
Austrian Case Study
Table 3 shows the compliance assessment results for Austrian monitoring data for 2006, obtained from the SWAD database. There are about 150 sites, but each site had been sampled multiple times over the year. At each site, pH, DOC, total water hardness, alkalinity and the total and dissolved concentration of copper, nickel and zinc was measured. The waters in this dataset have a high pH (mean = 8.0). Average DOC is 2.5 mg/l and total hardness averages 10.7 °dH. Overall, there are relatively few EQS failures for copper, nickel and zinc. For copper and zinc, an additional comparison is made between EQS failure rate using the the bio-met bioavailability tool and the existing EQS in Austria. These data show that the EQS failure rates after accounting for bioavailability are equal (for Cu) and higher (for Zn) than when bioavailability is not considered. However, the location of the exceedances changes (results not shown here) which ensures that sites at greatest risk are identified by use of a bioavailability -based approach.